|
Regulation and modulation of calcium entry and transmitter release in motor nerve terminals
We use microelectrode recordings of transmitter release, in combination with high-resolution calcium imaging in adult motor nerve terminals to examine the characteristics and modulation of the calcium entry that control transmitter release at the synapse. We have developed a method for imaging the spatial distribution of calcium entry following a single action potential stimulus. Using this approach, we have provided evidence that a very small subset of the available calcium channels opens in the nerve terminal with each stimulus. We hypothesize that transmitter release is triggered by the opening of single calcium channels in these nerve terminals and have begun to study the modulation of this process. We are interested in the mechanisms that control calcium entry and how this entry triggers transmitter release. |
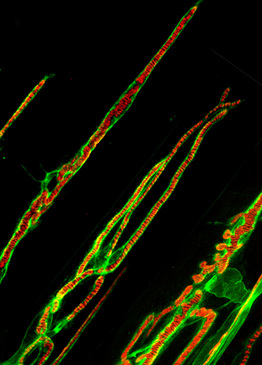
Frog neuromuscular junction stained with FITC peanut lectin and Alexa 594 conjugated a-bungarotoxin
|
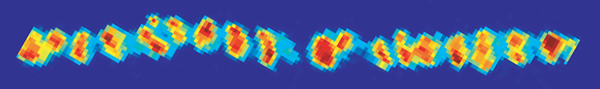
Average calcium entry into an adult frog motor nerve terminal during a single
action potential stimulation (average of 100 trials)
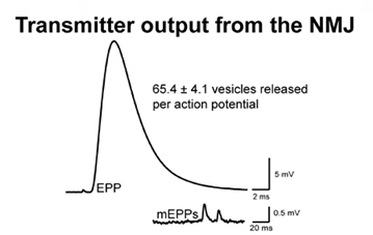
The magnitude of chemical transmitter released from a mouse neuromuscular junction recorded using and intracellular microelectrode. Quantal content is calculated by dividing the amplitude of the EPP by the average amplitude of mEPPs recorded from each synapse.
Microphysiological computer modeling
MCell computer models of calcium ion diffusion and binding reactions within the nerve terminal aid in the interpretation of data collected. Heavily constrained by over 70 years of anatomy, physiology, biochemistry, and molecular biology, realistic 3-D computer models of calcium ion diffusion and interaction within nerve terminals is modeled in collaboration with Dr. Markus Dittrich at the Pittsburgh Supercomputing Center.
MCell computer models of calcium ion diffusion and binding reactions within the nerve terminal aid in the interpretation of data collected. Heavily constrained by over 70 years of anatomy, physiology, biochemistry, and molecular biology, realistic 3-D computer models of calcium ion diffusion and interaction within nerve terminals is modeled in collaboration with Dr. Markus Dittrich at the Pittsburgh Supercomputing Center.

The image shows a frog neuromuscular junction stained using a red fluorescent label (Alexa 594-α-bungarotoxin) for postsynaptic receptors that accurately predict the location of active zones, and a green fluorescent label (FITC- peanut lectin) for the extracellular matrix around this synapse. The inset shows a screenshot rendered from an MCell reaction-diffusion computer simulation of calcium-triggered vesicle fusion in an active zone at this synapse (gray surfaces = nerve terminal membrane, large red spheres = docked synaptic vesicles, small black dots on the bottom of vesicles = calcium ion binding sites on synaptotagmin, small blue spheres = buffer-bound calcium ions, small yellow spheres = free ionic calcium, small red cylinders = closed voltage-gated calcium channels, small yellow cylinder = open voltage-gated calcium channels).
Transmitter release in control and disease model mouse motor nerve terminals
We have been using mouse neuromuscular preparations to study the regulation of transmitter release in both normal mice, and those that have been passively-transferred the disease Lambert-Eaton Myasthenic syndrome. In addition, we also use con-focal imaging of neuromuscular junctions stained with various antibodies directed against presynaptic proteins to characterize the presence and distribution of relevant molecules. This work furthers our understanding of calcium-dependent mechanisms, and is part of our effort to evaluate the effects of novel calcium channel agonists that might be of therapeutic benefit in diseases that result in neuromuscular weakness.
We have been using mouse neuromuscular preparations to study the regulation of transmitter release in both normal mice, and those that have been passively-transferred the disease Lambert-Eaton Myasthenic syndrome. In addition, we also use con-focal imaging of neuromuscular junctions stained with various antibodies directed against presynaptic proteins to characterize the presence and distribution of relevant molecules. This work furthers our understanding of calcium-dependent mechanisms, and is part of our effort to evaluate the effects of novel calcium channel agonists that might be of therapeutic benefit in diseases that result in neuromuscular weakness.
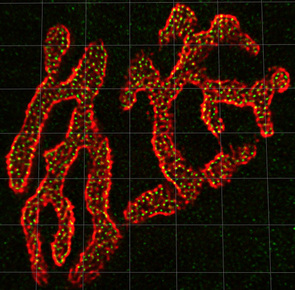
Confocal image of a mouse neuromuscular jucntion stained with Alexa-594 alpha-bungarotoxin (red) to label postsynaptic acetylcholine receptors on the muscle cell, and Alexa-488 labeled bassoon antibodies (green) to label synaptic vesicle docking sites (active zones) in the presynaptic nerve terminal. The average mouse neuromuscular junction has about 800 active zones that all roughly equally spaced from one another by about 500 nm. Grid lines = 5 uM
Development of novel calcium channel gating modifiers
We are collaborating with Dr. Peter Wipf's group in the Department of Chemistry to develop and test novel calcium channel gating modifiers that are selective for agonist effects on the types of calcium channels that regulate transmitter release at synapses (N- and P/Q-types). We use cell lines expressing various calcium channel subtypes as a model system to examine directly the gating and modulation of these channels, and the effects of various novel drugs. In particular, we are interested in novel drugs that increase the flux of calcium through open channels as potential first-in-class therapeutics for the treatment of neuromuscular diseases. A major focus of our studies is the disease Lambert-Eaton myasthenic syndrome that causes neuromuscular weakness due to anuto-antibody attack and removal of a fraction of the presynaptic calcium channels at neuromuscular junctions.
We are collaborating with Dr. Peter Wipf's group in the Department of Chemistry to develop and test novel calcium channel gating modifiers that are selective for agonist effects on the types of calcium channels that regulate transmitter release at synapses (N- and P/Q-types). We use cell lines expressing various calcium channel subtypes as a model system to examine directly the gating and modulation of these channels, and the effects of various novel drugs. In particular, we are interested in novel drugs that increase the flux of calcium through open channels as potential first-in-class therapeutics for the treatment of neuromuscular diseases. A major focus of our studies is the disease Lambert-Eaton myasthenic syndrome that causes neuromuscular weakness due to anuto-antibody attack and removal of a fraction of the presynaptic calcium channels at neuromuscular junctions.
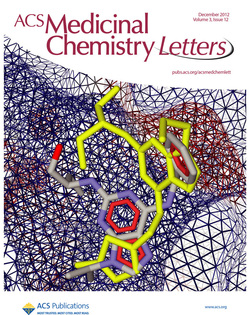
Docking of a novel calcium channel gating modifier to the cdk2/roscovitine complex. Our novel drugs were developed as derivatives of (R)-roscovitine and modified to reduce binding to cdk, while preserving calcium channel agonist activity. The electrostatic interaction surface at the binding site region is displayed and colored red for negative charge and blue for positive charge. Docking simulations were performed by Dr. Gabriela Mustata Wilson using Molegro Virtual Docker, taking into account side chain flexibility for all residues in the binding region.
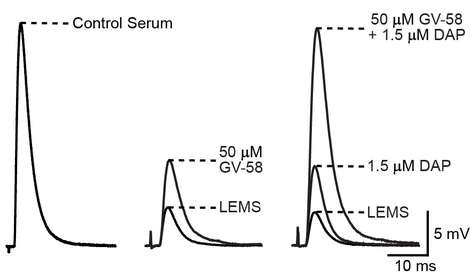
The supra-additive effect of GV-58 plus 3,4-DAP completely reverses the deficit in the magnitude of neurotransmitter release at Lambert-Eaton syndrome model mouse NMJs. Sample traces are displayed showing the average EPP amplitudes following exposure to the indicated conditions. Left: sample average EPP recorded from a NMJ of a control serum-treated mouse. Middle: sample average EPPs of a LEMS model NMJ before and after application of 50 M GV-58. Right: sample average EPPs recorded from a LEMS model NMJ before drug application (LEMS), following application of 1.5 µM 3,4-DAP, and following application of 50 µM GV-58 plus 1.5 µM 3,4-DAP. The combination of DAP plus GV-58 completely reverses neuromuscular weakness in this mouse model of Lambert-Eaton myasthenic syndrome.